| Giving Carbon Credit A brief series on the elemental substance of life Much is justifiably made about carbon and its impact on climate. But how much do you know about the much larger role carbon plays on our planet? You are what you think, and you are what you eat, and carbon is central to both. Carbon takes many forms, cycles through the environment, and is an integral part of our planet, especially the biosphere where all of our planet’s lifeforms live. Like the very air we breathe and the water we drink, carbon wears so many hats and takes on so many forms in our lives and in the life of this planet. The following short series of essays explores the myriad forms, movements and transformations of carbon through our planet and will provide additional resources if you want to find out more on your own. |
1. The Carbon Cycle
Forged in the heart of stars, carbon is the 4th most common element in the universe after hydrogen, helium and oxygen. Formed primarily by the triple fusion of 3 helium atoms in the heart of all stars, it has been scattered across the universe by exploding supernovas, whose remnant dust re-congeals into new stars and planets. With each generation of stars, the amount of carbon in the universe has increased, and scientists estimate that our sun and solar system has enough carbon in it to be in the third generation of stars that our galaxy has produced. Here on Earth, carbon is most commonly seen by itself in 3 forms: amorphous carbon/charcoal, graphite and diamonds. But its ability to bond into long chains with itself and other elements is what gives it the ability to create literally millions of different compounds. Run carbon through living organisms that combine it with hydrogen and you have hydrocarbons, the fuel of the industrialized world. Life processes—and some non-living chemical reactions—combine carbon with hydrogen and oxygen, and a handful of other elements, and that inexhaustibly complex world of organic chemistry is born. Life, of course wouldn’t exist without it.
“Geophysiology” is a term coined by James Lovelock by combining the words “geology” with “physiology.” This compound word has an almost carbon compound fruitfulness in its quest to explore the interaction of life forms with the geological processes of our planet. Geological processes are notably different here on Earth from what we see on other planets in our solar system, and by studying the metabolic inputs and outputs of populations of organisms and ecosystems, we have learned much about how life has altered those purely geological processes. Life has permeated the land, oceans and atmosphere on our planet, fundamentally altering the geological and chemical processes present in the “biosphere” along the way. Since carbon plays on both sides of the living/inanimate divide, the study of the carbon cycle is a central theme in the geophysiology of the Earth.
Many kinds of rocks and minerals have a “biogenic” component. For instance, limestone gets most of its calcium and carbon from diatoms and other shell bearing forms of life that chemically bond into the calcium carbonate basis of limestone. Many minerals, ranging from iron and uranium to coal and oil, were biologically concentrated to create the mineral ores, beds and deposits that we mine/pump from today. Furthermore, there are innumberable ways life is impacting geology in maintaining “geophysiological homeostasis.” Translation: just like your body maintains a certain level of oxygen in each cell in your body and keeps your temperature within a healthy range, the current mix of life on our planet has created conditions where our atmosphere is able to maintain a composition of 21% oxygen, where global temperatures are remarkably stable over time, etc. All of this despite there being global perturbations that should knock these numbers around much more than what we observe. In other words, the life systems have provided the geology of the planet a number of feedback mechanisms that keep the processes stable, much as your body keeps a more or less stable temperature/biochemical balance despite being out in the hot summer sun, being in cold windy weather, when you are physically active, inactive or sleeping, or when you have last eaten.
Lifeforms on the planet play central roles in many of the planet’s “thermostats” that maintain certain concentrations of gases, liquids temperatures, pH levels, etc. that happen to be conducive to the predominant life forms. These feedback mechanisms start with a supply of geologically dynamic substance such as carbon, which we will call a “pool,” out of which processes of life forms, geological, chemical and climatological forces pull out or deposit surpluses from/to that pool in order to maintain homeostasis. Movement in and out of these “pools” is called “flux:” if the net movement is into a pool, the pool is called a “sink.” If the net movement is out of the pool, it is called a “source” of that substance. Sinks and bathtubs are appropriate metaphors for these feedback loops, which accurately describes how the level of the “water” depends on how well the water coming in through the faucet(s) is matched by the water leaving through the drain(s).
For example, plants can be a carbon “sink” when plants grow biomass, pulling from the soil and atmospheric pools of carbon during the growing season. During winter, plants switch roles, releasing carbon back into those atmospheric and soil pools as dormant vegetation decomposes and biomass production stops, turning plants from a net carbon sink into a net carbon source in relation to the atmospheric and soil carbon. Wet, warm weather increases carbon uptake from plants, and either hot or cold, dry weather can switch plants into net carbon sources. The famous Keeling curve that charts northern hemisphere atmospheric CO2 concentrations reflects this shifting role, with the concentration of atmospheric CO2 peaking right before growing season kicks in in April, later bottoming out in August, when CO2 plant absorption peaks and begins to taper off as fall begins, leading into winter.
The carbon cycle can be seen as having two large, interconnected complexes, however. Plants are part of what is called the “fast carbon cycle,” which includes phytoplankton in the ocean, microorganisms in the soil, and pretty much all other living organisms that swap carbon with each other and the immediate environment. It is called the “fast carbon cycle” because carbon cycles through these pools relatively quickly. You can follow carbon through the fast carbon cycle beginninng with when plants pull carbon dioxide from the atmosphere and use solar energy to convert it into sugars and build biomass, which becomes the food source for the rest of the ecosyem. Plants are the “producers” that feed the animals who eat plants, who in turn are eaten by other animals. Animals are called “consumers” in this energy transferring pyramid, joined by the “decomposers” who break down living tissues into components that can be used to build new biomass in the form of new plants, animals, microbes, etc. Carbon is swapped rather quickly in this fast carbon cycle, moving along one lifetime at a time, as lifeforms live and die, eat and are eaten or decompose after dying.
The “slow carbon cycle” is the other complex that, while being intimately connnected to the life-based fast carbon cycle, operates on a much larger and slower scale. In contrast to the relatively rapid cycling of carbon through living biomass, the planet cycles carbon through the rocks, soil, the ocean, and the atmosphere at a much more leisurely pace of 100-200 million years. Carbon erodes from the land and settles out of the ocean waters, desposited into sedimentary layers that turn into rock, which is subducted under the continental plates, only to melt and re-emerge via volcanic eruptions and mountain building/wearing away, to begin the cycle again. Carbonic acid is formed when rain absorbs carbon from the atmosphere, and this mild acid dissolves rock in a process called chemical weathering. Limestone and other sedimentary rock forms on the ocean bed when the carbon and calcium from microorganisms and from chemical weathering bonds with calcium and carbonate ions in the ocean water. Similarly, coal beds and oil bearing formations were formed during periods of the Earth’s history when there was enough biomass being grown to build up and be compressed/concentrated into carbon-rich deposits we call fossil fuels.
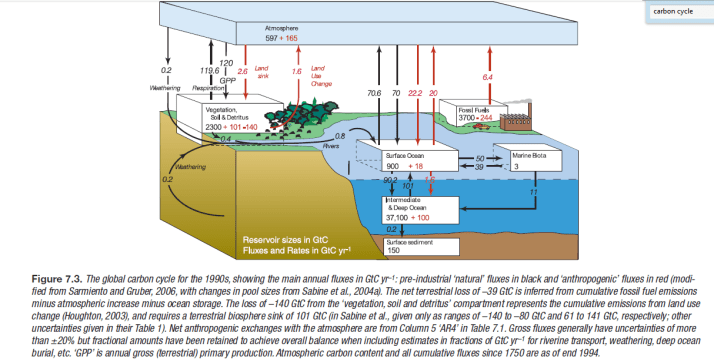
This depiction of the carbon cycle is reproduced from the 2007 IPCC AR4 Working Group on the Carbon Cycle and the Climate System, Chapter 7.3
More resources for further exploration:
The Carbon Cycle
The Carbon Dioxide Information Analysis Center